NORA PHILBIN, BA
Institute of Neurology
Queen Square, London WCIN 3BG
INTRODUCTION
Stereotypic behaviour is an abnormal behaviour frequently seen in laboratory primates. It is considered an indication of poor psychological well-being in these animals. As it is seen in captive animals but not in wild animals, attention has been focused on the situations in which this behaviour develops. However, the emphasis of research has been on reducing or eliminating established stereotypic behaviour in laboratory primates. The focus of this paper will be to highlight possible factors contributing to the development of stereotypic behaviour in laboratory primates, specifically macaques, by examining the relevant scientific literature. Stereotypic behaviour will be defined, its use as a marker of welfare will be investigated, and experimental evidence as to the possible causes of the behaviour will be reviewed.
What is stereotypic behaviour?
Stereotypic behaviour has been defined as a repetitive, invariant behaviour pattern with no obvious goal or function.1 A wide range of animals, from canaries2 to polar bears3 to humans4,5,6 can exhibit stereotypes. Many different kinds of stereotyped behaviours have been defined and examined. Examples include crib-biting and wind- sucking in horses,7 eye-rolling in veal calves,8 sham-chewing in pigs,9 and jumping in bank voles.10 Stereotypes may be oral or involve bizarre postures or prolonged locomotion.
A good example of stereotyped behaviour is pacing. This term is used to describe an animal walking in a distinct, unchanging pattern within its cage. The walking can range in speed from slow and deliberate to very quick trotting. It may involve only a few circuits or it may be prolonged, lasting several minutes. The locomotion may be combined with other actions, such as a head toss at the corners of the cage, or the animal rearing onto its hind feet at some point in the circuit. The pattern and appearance of the stereotype differs from animal to animal. Stereotypes often go through stages of development. In the early stages, the behaviour may be easily interrupted by a loud noise or other stimulus. At later stages, such interruption is difficult or impossible. At this stage, stimuli can even escalate performance of the behaviour. The animal can appear to be in a trance-like state, disconnected or detached from its surroundings.1
The distinction between stereotypic and non-stereotypic behaviour can be illustrated by considering eating behaviour. An animal may eat in a set manner, sitting in the same place, using a distinct motor pattern for a prolonged time but this would not be considered abnormal stereotyped behaviour as it has an obvious function. Other abnormal behaviours such as severe aggression or inappropriate/inadequate maternal behaviour cannot be considered stereotypic because they are not repetitive and invariant or ritualised. Stereotypic behaviour is evaluated in terms of frequency and duration, in other words, how often does the animal engage in bouts of such behaviour and how long do the bouts last? It is not adequate, therefore, simply to say that an animal shows stereotypic behaviour; it is necessary to quantify the levels of the behaviour exhibited. This is usually done by examining the animal's time budget. There may be less concern for an animal that spends 3% to 5% of its time engaged in stereotypic behaviour than one that spends 75% of its time in stereotypy. However, it can be argued that any amount of stereotypy is a cause for concern and is an indication of poor welfare.
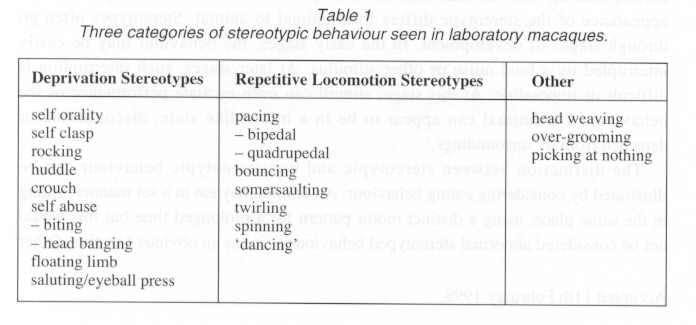
Stereotypes in non-human primates
The stereotypes of non-human primates are generally divided into two categories: deprivation stereotypes and cage stereotypes,11,12 (see Table 1). Deprivation stereotypes are also referred to as self-directed stereotypes because the behaviours are performed on the individual's own body. For example, self orality describes non- nutritive sucking of an animal on one or more parts of its body (e.g. fingers, tail, or genitalia). Rocking, huddle, and crouch are abnormal postures. Self abuse refers to self injurious behaviour, such as biting, scratching, or head banging. The term floating limb is used to describe a phenomenon which involves an animal's limb appearing to move of its own accord. This surprises the animal which will then threaten and attack the limb. A salute is another abnormal posture which in12volves the animal placing a hand in front of its face, palm out, with one or more fingers pressed against the eyeball. Deprivation stereotypes are usually seen in monkeys that have been separated from their mothers at birth or within the first year of life and raised in part or total social isolation13,14,15,16 These abnormal behaviours are thought to be analogous to the normal behaviours seen in infant and juvenile monkeys. Here however they become self-directed in the absence of a con-specific. Self orality in isolates, specifically self- sucking of digits or tail, is considered to be analogous to nipple sucking in infants raised with their mothers. Self clasp is related to mother clinging or clasping. Self abuse is considered to be the only outlet for these animals' frustration.
Repetitive locomotion stereotypes include pacing, jumping in place, and somer-saulting13 (see Table 1). As the name implies, these cage stereotypes are considered to be the result of the living environment of an animal. All of these stereotypes are active, involving dynamic, whole body movements. Dancing refers to a back and forth quadrupedal movement which is distinct from pacing or spinning.
Though helpful, these terms do not encompass all forms of stereotypic behaviour nor do they completely eliminate confusion regarding the different forms. Some behaviours are not included in either category .Stereotypes such as these are placed in v a third miscellaneous category (see Table 1). For example, head weaving, which refers to a head toss which is not involved in a pacing stereotype. Also in this category is the behaviour referred to as over-grooming, where an animal removes all the hair from one part of its body, usually a small patch on the shoulder or arm. The third stereotype in this category is picking at nothing (self explanatory).
Different authors define stereotypic behaviour differently. They may observe different kinds of behaviour and categorise these behaviours as they see fit. For example, Paulk et al. (1977),17 use the term walk in circles to describe an animal pacing completely around its cage and the term pace to describe an animal pacing up and down only one side of its cage. Berkson13 on the other hand, refers only to pacing with no such distinction. The term twirling is also defined differently by Paulk et al. (1977)17 who refer to it as partially bipedal walking, than it is by Capitanio (1986)12 who refers to it as a stationary quadrupedal movement. Unfortunately, this can and does lead to confusion and disagreement as to what is and is not a stereotyped behaviour. This report will focus primarily on repetitive locomotion stereotypes in laboratory macaques. For the purpose of clarity , the term stereotypic behaviour when used here will refer to characteristically fixed motor activities which are repetitively performed by an individual animal.
Which laboratory primates exhibit stereotypic behaviour?
Repetitive locomotion stereotypes are found in many common laboratory primates, including marmosets, squirrel monkeys, and several species of macaque. In their paper on stereotypic behaviour in marmosets, Berkson and his colleagues (1966)18 describe the effects of isolation rearing in a germ-free environment on two marmosets. They concluded that marmosets, in contrast to macaques, do not develop deprivation stereotypes in these rearing conditions. However, they do mention two juvenile animals in the colony that showed repetitive locomotion stereotypes. Squirrel monkeys display deprivation stereotypes and repetitive locomotion stereotypes when separated from their mothers at birth and nursery reared in individual cages!9 These animals showed self orality, rocking, and huddling (equivalent to self clasping), as well as a pacing-Iike stereotype referred to as looping and a locomotion stereotype termed rolling-in-a-ball. These authors found the occurrence of stereotypic behaviour in these animals was higher than in the marmosets but lower than in macaques. Capitanio's (1986)12 review of behavioural pathology in primates highlights work by several authors on repetitive locomotion stereotypes in macaques. These studies indicate that stump-tail (M. arctoides), long-tail (M. fascicularis), Japanese (M. fuscata), and rhesus (M. mulatta) macaques all exhibit repetitive locomotion stereotypes. Most of the studies involved animals that were reared in part or total isolation. This is significant because most laboratory macaques would not be reared under such restrictive conditions. However, the study of stump-tails was designed to investigate the abnormal behaviour of captive bred but not isolated animals.
Stereotypic behaviour and animal welfare
As this behaviour is seen in several species of laboratory primates, it has become the focus of concern regarding the welfare of animals that show it. As stated above, stereotypic behaviour is considered to be indicative of poor psychological well-being and poor general welfare. The use of stereotypic behaviour as a marker of welfare is well established and can be easily illustrated.
The UK Code of Practice for the Housing and Care of Animals in Scientific Procedures (1989)20 states that animals must be maintained in good health and physical condition, behaving in a manner normal for the species and strain. Stereotypic behaviour is not seen in animals in the wild and is understood to be abnormal. Therefore, in the UK, it is considered a symptom of poor welfare. The same is true in the United States. The US Animal Welfare Act amendment of 1985 (published in 1991)21 required researchers to promote the psychological well-being of the captive primates in their care. This is hampered by the lack of an effective definition of this term. Scientists in the US have commented that without a proper definition it is nearly impossible to construct a program to promote psychological well-being or to recognise its absence.22,23,24 Unfortunately, a useful measure of psychological well-being remains elusive. It is difficult to ascertain in humans, let alone in monkeys. A great deal of debate and controversy has surrounded what does and does not constitute psychological well-being in primates. However, in 1993 the International Primatological Society proposed that stereotypes should be used as a behavioural indicator of poor psychological well-being.25 As far as this organisation is concerned stereotypes are serious enough to be considered a welfare problem. Beyond the formal regulations on animal welfare are the conclusions drawn from scientists who have investigated stereotypic behaviour. Mason (1991) concludes in her review of stereotypes,' that stereotypic behaviour is indicative of an inadequate and possibly aversive environment. She is equivocal as to whether or not stereotypic behaviour represents a welfare problem. Broom (1987), however, comments that the reduced responsiveness seen in sows exhibiting stereotypes is an indication of poor welfare.26 These comments are echoed in the statements of animal rights campaigners. Wemelsfelder (1993)27 states in her discussion of animal boredom and its relationship to stereotypic behaviour that "the development of stereotypic behaviour reflects an impairment of an animal's capacity to interact with the environment." In her opinion "such a process may be regarded as direct evidence of chronic suffering". Roger Fouts remarks in his book, Next of Kin, that stereotyped pacing in laboratory macaques is a "sign of severe stress" in these animals.28
From this evidence it is clear that stereotypes are used as markers of animal welfare within the scientific community. An emphasis on the current living environment of the animal is apparent. Not surprisingly, research into the factors underlying stereotypic behaviour have focused on environmental causes.
Possible environmental causes of stereotypic behaviour
In the search for causes, the emphasis historically has been placed on an animal's environment. It has been believed that stereotypic behaviours will develop in a deficient environment.12,17 Several aspects of the captive environment of laboratory primates have been investigated. These include cage size, housing type (individual vs pairing or groups), stress, and lack of environmental complexity.
Belief in the need to sterilize laboratory caging has led, in the past, to the use of small cages which could be cleaned in conventional cage wash machines. Use of such small cages has long been implicated as a cause of stereotypic behaviour. The belief is that wild animals confined in small cages will develop abnormal stereotyped behaviour.9 The main aim of specification of minimum cage sizes per body weight of animal was to promote more species-specific behaviour (including the vertical flight reaction) and to reduce abnormal behaviours. The International Primatological Society (1993) considers size of enclosure as one of four significant physical factors concerning the housing of non-human primates.25
The type of housing is an environmental factor that has been linked to stereotypic behaviour in laboratory primates. In the past, laboratory macaques were housed individually to reduce the risk of wounds due to fighting and the spread of infection. In recent times, social housing in pairs or groups whenever possible has been favoured over individual housing.'" Primates are social animals and normally live in large groups in the wild. Individual housing, even with auditory, olfactory, and visual contact with con-specifics, has been considered by some to be akin to solitary confinement. Thus individual housing has been thought to contribute to development of stereotyped behaviours.
Stress is another factor of concern. It has been suggested that the stress of life in a laboratory might contribute to the development of abnormal behaviours. Laboratory primates experience a plethora of stressors, potentially including injections, blood drawing, cage changes, physical and chemical restraint, and participation in scientific procedures. The inability of the animal to escape from these experiences might lead to adoption of stereotyped behaviours as a means of coping with an aversive environment. Some studies have indicated that stereotypic behaviour produces a 'trance-like' state31 and is linked to reduced awareness of the environment! Kravitz et at. (1980)32 associate stereotypes in human children with analgesia.
One final aspect of the environment that has been considered to cause stereotypic behaviour is boredom. Primates are intelligent, active, curious animals with a need for complex, stimulating environments. Primates in laboratories are considered especially at risk of being bored. The large amount of research into the development of enrichment devices for primates in laboratories and the effects of these interventions on stereotypic behaviour reflects this belief.33,34,35
Research into the "causes" of stereotypic behaviour
Much of the research into the causes of stereotypic behaviour has focused on cage size, perhaps because it is the most easily manipulated variable of the four mentioned above. Small cage size seems intuitively to be a likely candidate for causing harmful effects in laboratory primates. One of the oldest and most often cited studies on cage size is the work of Draper and Bernstein (1963).29
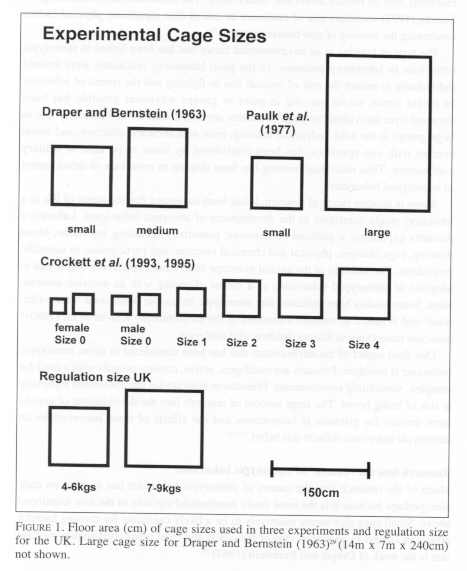
In this experiment, the behaviour of twelve wild-born three year old rhesus monkeys was evaluated in three different cage sizes (see Figure 1). The largest cage size used was more akin to a housing pen than a cage. The results state that any stereotypic behaviour seen in the smallest cage, was significantly reduced in the medium sized cage, and was not observed in the largest cage. The authors used as an example a female monkey which exhibited continuous backward somersaults in the small cage. In the medium cage this animal showed stereotyped pacing, occasionally throwing up her forelegs and tossing her head back. The authors interpreted this to be the beginning of a somersault which was not completed. In the largest cage, this animal was not observed engaging in stereotypic behaviour. The authors concluded housing monkeys in small cages leads to development of stereotypic locomotion as a substitute for normal locomotion. Once the spatial restriction had been removed then normal locomotion resumed.
Paulk et at. (1977) found similar links between cage size and stereotypic behaviour17 (see Figure 1). In this experiment twenty-four rhesus monkeys, both wild- born and captive bred, were examined in a small and a large cage. The results of this study indicate that, in the eight monkeys that showed stereotypic behaviour, stereotypic locomotion increased in the smaller cage while normal locomotion decreased. The authors state in the discussion that prolonged housing in small cages leads to stereotyped locomotion. This conclusion is based presumably on the length of time each monkey had spent in a small cage. The animals which exhibited stereotypes were older and had spent more time in small individual cages.
More recent work by Crockett and others (1993, 1995)36,37 has come to different conclusions. This study investigated the effect of cage size on urinary cortisol levels36 and behaviour37 in twenty long-tail macaques. All the animals were wild-born, captured and imported as adults. Five cage sizes were used in this experiment (see Figure 1). After 48 hours, the most restrictive cage (Size 0) was slightly enlarged to allow the animals to move more freely. This cage size was also larger for the males than for the females. All of the cages used were substantially smaller than the ones used in either of the two experiments discussed above. Furthermore, all of the cages are smaller than the legal minimum cage size for animals of this weight in the UK (89 cm x 89 cm x 110 cm for a 4-6 kg animal; see Figure 1).
After a two-week preparation phase in which the animals habituated to the experimental room, the monkeys were systematically moved from one cage size to another every two weeks until each monkey had experienced each cage size. The first cage size was assigned at random but the order was the same for each monkey (0-1-4-2-3). Each monkey experienced the same changes of cage sizes, both increases and decreases. The experiment phase lasted ten weeks. Two and one half years later the monkeys were evaluated behaviourally in a follow-up phase.
Urinary cortisol responses as well as behavioural data were gathered. An elevation in levels of urinary cortisol is usually accepted as an indicator of stress in animals. Urinary cortisol levels were unrelated to cage size and no significant elevation in cortisol levels were seen in any of the cage sizes. The results also indicated that abnormal behaviour did not differ significantly across cage sizes. The conclusion drawn from the cortisol responses was that variations in cage size have no stress effect as judged by urinary cortisol. The conclusion drawn from the behavioural data was that cage size was the least important of all independent variables investigated. Some of the other variables examined were phase of the experiment, subject, sex and days in captivity.
These results do not stand alone. They agree with the results found for pig-tail macaques (M. nemestrina) in a similar experiment conducted by this group,38,39 as well as to those found by Goosen (1988)40 in his exploration of cage size and stereotyped locomotion. Likewise, the research by Line et at. (1990)41 investigating the differences in heart rate and behavioural patterns in relation to cage size in female rhesus monkeys. These authors found that increased cage size had no significant effect on behaviour, heart rate, or activity levels.
Furthermore, Crockett et al. (1995)37 state that they found no evidence that long-tail macaques taken from the wild as adults develop substantial levels of abnormal behaviour .This was the case, even though these animals were housed individually for more than three years in laboratory caging with minimal provision of social contact and enrichment devices. This is an example of macaques experiencing all the possible environmental "causes" of stereotypic behaviour including small cages, individual housing, the stress of experimental manipulation and presumably boredom. Yet these monkeys did not develop abnormal or stereotyped behaviour. This seems to indicate that the causes of stereotypic behaviour are not as straightforward as previously described.
Considering the conflicting experimental evidence it is likely that there may be other complicating factors contributing to the development of stereotypic behaviour in laboratory macaques. Crockett et al. (1995)37 suggested a few areas of interest to be explored, namely, rearing history , breed and genotype of the animals, and differences in individual disposition. Each of these topics will be discussed in turn.
Complicating factors - rearing history
It is apparent from isolation studies that animals separated from their mothers at birth and raised in partial or total social isolation develop deprivations stereotypes.13,14,15,16 However, it is interesting to note the rearing histories of the animals observed in the studies discussed previously. The animals used by Crockett et al. (1993, 1995)36,37 which failed to develop significant levels of stereotypic behaviour, were all wild-born animals who were captured and imported as adults. The female rhesus monkeys in the heart rate study by Line et al. (1990)41 were reared in social housing until approximately twenty months of age. Of the eight animals that showed stereotypic behaviour in the Paulk et al.(1977)17 cage size experiment, six had been raised in a laboratory and separated from their mothers at three months of age. The animals in the Draper and Bernstein (1963)29 cage size study were three years old at the time of the experiment and had been in the laboratory for two years. This indicates, presumably, that they had been captured and separated from their mothers when they were less than one year old. The nursery-reared squirrel monkeys in Roy (1981)19 experiment were separated from their mothers at one day after birth. Furthermore, the maternally reared controls in that experiment did not develop stereotypic behaviour. Therefore, it is possible that the rearing histories of these animals contributed to the development of their stereotypic behaviour.
A paper by Marriner and Drickamer (1994)42 on the factors influencing stereotypic behaviour in primates in a zoo also highlights the effect of rearing. The animals in their study which showed the highest level of stereotypic behaviour were those that had been hand-reared by humans. They comment that "in our study hand-reared animals performed significantly more stereotypic behaviour than mother reared animals even though hand and mother reared animals were born in the same year and exposed to the same changes and improvement in the zoo environment". They highlight in their conclusions that stereotypic behaviour is probably influenced by the rearing history of an animal.
In his paper on prevention of abnormal behaviour in rhesus monkeys, Goosen (1988)40 recommends that infants be weaned only when they are three to six months old. He observed no stereotypic locomotion in nursing infants but did observe "pre-stereotypic" locomotion in weaned juveniles (both individually and social housed). He describes this "pre-stereotypic" locomotion as agitated and non-functional but lacking the fixed appearance of true stereotypic pacing. Goosen observed this "pre-stereotypic" shortly after weaning in these animals. The author concludes that stereotypic locomotion is a response to early maternal separation in these animals.
An additional example of the possible effect of rearing history on abnormal behaviour includes the work done by Wesseling (1988)43 at the Primate Centre TNO in the Netherlands. This study examines faeces smearing (which the authors termed "monkey graffiti") by laboratory macaques. This behaviour, though not stereotypic because it is not performed ritualistically or in repetition, is considered abnormal. The behaviour of both wild-born and laboratory-born animals was examined. The captive- born animals had been weaned at three months and housed together in groups until two years of age at which time they were moved to individual housing. Most of the wild-born animals were older and therefore had been captive and in individual housing longer than the laboratory-born animals. Over half of the laboratory-born animals showed this abnormal behaviour while none of the wild-born animals did. Furthermore, ten monkeys that had been born in a safari park and subsequently moved to the centre when they were older than one year did not show this abnormal behaviour .
From this evidence, it seems that the rearing history of an animal will influence whether or not that animal will show abnormal behaviour. The longer an animal stays with its mother, the less likely it seems to be that it will develop abnormal or stereotyped behaviour .
Breed and genotype of the animal
It is apparent from the literature that different species of macaques will respond differently to the same stressful stimuli or situation. Several papers have shown this to be the case in experimental settings. Clark et at. (1994)44 investigated the heart rate patterns of rhesus, bonnet, and long-tail macaques under three stressful experimental conditions. These were routine husbandry procedures in the animals' home cage, introduction into a novel environment and full physical restraint. Overall mean heart rate across the three experimental conditions was lowest in the rhesus, intermediate in bonnets and highest in the long-tails. These authors conclude that the three species differ in their behavioural and psychophysiological responses to stressful situations.
Boccia et at. (1995)45 also found profound differences between species in a study comparing behaviour of bonnet and pig-tail macaques. In this experiment, six groups of macaques underwent twenty-four hour water deprivation followed by access to a single water spout. Animals usually line up according to rank and wait their turn to drink from the spout. This technique is often used to determine rank in large groups of monkeys. The pig-tail macaques showed more aggression and antagonistic behaviour around the water spout than did the bonnets. They competed vigorously for access to the spout with lots of pushing and shoving. In contrast, the bonnets seemed simply to queue up in dominance order and wait quietly for their turn at the water spout. A striking aspect of Capitanio's (1986)12 review of repetitive locomotion stereotypes is the lack of data for pig-tail macaques. He has no reference for the investigation of this type of stereotypic behaviour in pig-tail macaques. There are, however, anxiety responses as infants also displayed the largest response to stressful stimuli as young adults. Furthermore, animals which were paternally related (half-siblings) tended to have similar anxiety levels. The authors conclude "it seems likely that there are inborn or constitutional differences among individual monkeys in their 'threshold' for anxiety and that these differences might :well have a genetic or prenatal basis".
Boccia et at. (1995)45 also investigated the intrinsic factors that influence stress responses in macaques. These authors looked at the individual behaviour of both pig- tail and bonnet macaques during a potentially stressful event: exiting the home pen to a smaller cage from which they would be removed for blood drawing. They found that older animals entered the transfer cage and volunteered for blood drawing more readily than younger animals. They concluded that factors such as a familiarity with and cognitive understanding of the experimental protocol (sooner in-sooner out) led to lower anxiety in these animals. Animals which were intellectually capable of interpreting what was required of them were less stressed by the experience. Furthermore, the authors propose that individual animals will react to stressful events differently depending on their temperament. Studying infant pig-tail macaques they determined that animals with high baseline heart rates showed more pronounced behavioural and physiological reactions to maternal separation than did animals with lower baseline heart rates. They conclude that individual differences between animals should be considered when designing welfare and research protocols.
CONCLUSIONS
By examining the scientific literature regarding stereotypic behaviour in laboratory macaques, it is possible to move towards an understanding of this abnormal behaviour. Experimental evidence seems to indicate that environmental factors such as cage size, housing type, stress and boredom which are often cited as the "causes" of stereotype in laboratory animals, do not necessarily cause the behaviour. Not all monkeys that experience these environmental conditions develop stereotypic behaviour . However, in animals which already exhibit the behaviour, these conditions will tend to accentuate it. Thus, these environmental components appear to influence the frequency of established stereotypic behaviour but do not cause it.
There is experimental evidence to suggest that intrinsic factors determine whether or not an individual animal will develop stereotypic behaviour. Factors such as rearing history, breed or genotype, and individual disposition will all contribute to the psychological makeup of an animal. Animals which are separated from their mothers early in life seem to be more prone to developing abnormal or stereotypic behaviour. The different breeds of macaques respond differently to stressful stimuli and situations, including maternal separation. Finally, an animal's individual personality appears to contribute to the likelihood of it exhibiting stereotypic behaviour.
Stereotypic behaviour is believed to signify poor psychological well-being and constitute a welfare problem in laboratory macaques. However, this behaviour is extremely complex and is not completely understood in animals or humans. Although it cannot be argued that stereotypic behaviour is not a welfare problem, this author believes that it is not enough simply to conclude that stereotypic behaviour is a sign of poor welfare in laboratory macaques. That conclusion, in the absence of a fundamental understanding of the behaviour, may lead to condemnation of research - facilities, animal care providers, and biomedical research in general. None of these may be directly responsible for the development of stereotypic behaviour in laboratory primates. It is crucial to determine why these animals exhibit stereotypic behaviour and to address the underlying causes, which may relate to experiences years before. Only in this way can we move towards an effective method of completely eliminating this behaviour in laboratory macaques.
Acknowledgments
I would like to thank Prof. R. Lemon for his support and encouragement as well as Dr S. Baker and Miss R. Spinks for useful discussions and comments on the manuscript.
References
1. Mason, GJ. (1991). Stereotypies: a critical review. Animal Behaviour 41: 1015- 1037.
2. Keiper, R. (1970). Studies of stereotypy function in the canary (Serinus canarisus). Animal Behaviour 18:353-357.
3. Wechsler, B. (1991). Stereotypies in polar bears. Zoo Biology 10:177-18.
4. Butt, C. and Butt, SJ. (1965). The effects of environmental complexity on the stereotyped behaviour of children. Animal Behaviour 13: 1-4.
5. Thelen, E. (1979). Rhythmical stereotypies in normal human infants. Animal Behaviour27: 699-715. 6. 6. Soussignan, R. and Koch, P. ( 1985) .Rhythmical stereotypies (leg-swinging) associated with reductions in heart rate in normal school children. Biological Psychology 21:161.
7. Kennedy, MJ., Schwabe, A.E. and Broom, DM. (1993). Crib-biting and wind- sucking stereotypies in the horse. Equine Veterinary Education 5(3):151-154.
8. Fraser, A.F. and Broom, D. (1990). Farm animal behaviour and welfare. London: Bailiere Tindall. 9. Broom, D.M., Mendl, M.T. and Zanella, AJ. (1995). A comparison of the welfare of sows in different housing conditions. Animal Science 61:369-385.
10. Odberg, R. (1986). The jumping stereotypy in the bank vole (Clethrionomys glareolus). Biological Behaviour 11:130-143.
11. Berkson, G. (1967). Abnormal stereotyped motor acts. In comparative psycho-pathology: animal and human. Ed. by J. Zubin & H.F. Hunt, Chap. 4:76-94.
12. Capitanio, J.P. (1986). Behavioural pathology. In comparative primate biology: behaviour, conservation, and ecology. Ed. by G. Mitchell & J. Erwin, 2A:411- 454.
13. Berkson, G. (1968). Development of abnormal stereotyped behaviors. Develop- mental Psychobiology, 1:118-132.
14. Suomi, SJ., Harlow, H.F. and Kimball, S.D. (1971). Behavioral effects of prolonged partial social isolation in the rhesus monkey. Psychological Reports 29:1171-1177.
15. Anderson, J.R. and Chamove, AS. (1980). Self-aggression and social aggression in laboratory-reared macaques. Journal of Abnormal Psychology, 89: 5391-1550.
16. Sackett, G.P ., Ruppenthal, G.C., Fahrenbruch, C.E., Holm, R. and Greenough, W.T. (1981). Social isolation rearing effects in monkeys vary with genotype. Developmental Psychology 17:313-318.
17. Paulk, H.H., Dienske, H. and Ribbens, L.G. (1977). Abnormal behavior in relation to cage size in rhesus monkeys. Journal of Abnormal Psychology 86:87-92.
18. Berkson, G., Goodrich, J. and Kraft, I. (1966). Abnormal stereotyped move- ments of marmosets. Perceptual and Motor Skills 23:491-498.
19. Roy, M.A. (1981). Abnormal behaviors in nursery-reared squirrel monkeys (Saimiri sciureus). American Journal of Primatology 1 :35-42.
20. Home Office/British Government (1989). Code of practice: for the housing and care of animals used in scientific procedures. Presented pursuant to Act Eliz. II 1986 C.14 Section 21 (Animals (Scientific Procedures) Act 1986), Section 3.2 p12.London.
21. U .S. Department of Agriculture (1991). Animal welfare standards, final rule (Part 3, subpart D: specifications for the humane handling, care, treatment, and transportation of nonhuman primates). Federal Register 56:6495-6505.
22. Gray Eaton, G., Kelley, S.T., Axthelm, M.K., Iliff-Sizemore, SA. and Shiigi, S.M. (1994). Psychological well-being in paired adult female rhesus (Macaca mulatta).American Journal of Primatology 33:89-99.
23. Novak, MA. and Suomi, S.J. (1988). Psychological well-being of primates in captivity. American Psychologist 10:765-773.
24. Vandenbergh, J.G. (1989). Issues related to "psychological well-being" in nonhuman primates. American Journal of Primatology Supplement 1:9-15.
25. International Primatological Society (1993). IPS international guidelines: IPS code of practice: 1: Housing and environmental enrichment. Primate Report 35: 8-16.
26. Broom, D. (1987). Applications of neurobiological studies to farm animal welfare. In Biology of stress in farm animals: an integrated approach. Ed. by P .R. Wiepkema and P.W.M. van Adrichem. 42:101-110.
27. Wemelsfelder , F. ( 1993) .The concept of animal boredom and its relationship to stereotypic behaviour. In Stereotypic animal behaviour: fundamentals and applications to welfare. Ed. by A.B. Lawrence and J. Rushen. Chap. 4:65-95.
28. Fouts, R. (with Stephen Tukel Mills) (1997). Next of kin: what chimpanzees have taught me about who we are. 1st ed., p309. William Morrow and Company Incorporated, New York.
29. Draper, W A. and Bernstein, IS. (1963). Stereotyped behaviour and cage size. Perceptual and Motor Skills 16:231-234.
30. Reinhardt, V. (1994). Pair-housing rather than single housing for laboratory rhesus macaques. Journal of Medical Primatology 23:426-431.
31. Stevenson, M.F.(1983). The captive environment: its effect on exploratory and related behavioural responses in wild animals. In Exploration in animals and man. Ed. by J. Aecher & L.I.A. Birke. pp 176-197. 32. Kravits, H., Rosenthal, V ., Teplitz, Z., Murphy, J.B. and Lesser, R.E. (1980). A study of head-banging in infants and children. Disorders of the Nervous System 21 :203-208.
33. Bayne, K., Mainzer, H., Dexter, S., Campbell, G., Yamada, F. and Suomi, S. (1991). The reduction of abnormal behaviors in individually housed rhesus monkeys(Macaca mulatta) with a foraging/grooming board. American Journal of Primatology 23:23-35.
34. Schapiro, SJ., Bloomsmith, M.A., Suarez, S.A. and Porter, L.M. (1990). Effects of social and inanimate enrichment on the behavior of yearling rhesus monkeys. American Journal of Primatology 40:247-260.
35. Schapiro, SJ. and Bloomsmith, M.A. (1994). Behavioral effects of enrichment on pair-housed juvenile rhesus monkeys. American Journal of Primatology, 32:159-170.
36. Crockett, CM., Bowers, CL., Shimoji, M., Bellanca, R. and Bowden, DM. (1993). Urinary cortisol responses of long-tailed macaques to five cage sizes, tethering, sedation, and room change. American Journal of Primatology 30:55-74.
37. Crockett, C.M., Bowers, C.L., Shimoji, M., Leu, M., Bowden, D.M. and Sackett, G. (1995). Behavioral responses to long-tailed macaques to different cage sizes and common laboratory experiences. Journal of Comparative Psychology Vol. 109(4): 368-383.
38. Crockett, CM., Bowers, CL., Shimoji, M., Bellanca, R. and Bowden, DM. (1994). Behavioral responses to four cage sizes of home cage by adult female pig-tailed macaques. American Journal of Primatology 33:203-204. (Abstract).
39. Crockett, C.M., Bowers, C.L., Shimoji, M., Leu, M., Bellanca, R. and Bowden, DM. (1993). Appetite and urinary cortisol responses to different cage sizes in female pig-tailed macaques. American Journal of Primatology 30:305. (Abstract).
40. Goosen, C. (1988). Developing housing facilities for rhesus monkeys: prevention of abnormal behaviour. In New developments in bioscience: their implications for laboratory animal science. Ed. by A.O. Beynen and H.A. Solleveld. Martinus Nijhoff Publishers. pp 67-69.
41. Line, S.W., Morgan, K.N., Markowitz, H. and Strong, S. (1990). Increased cage size does not alter heart rate or behavior in female rhesus monkeys. American Journal of Primatology 20:107-113.
42. Marriner, LM. and Drickamer, L.C. (1994). Factors influencing stereotyped behavior of primates in a zoo. Zoo Biology 13:267-275.
43. Wesseling, D., Baumans, V. and Goosen, C. (1988). Monkey graffiti (environmental deficiency, boredom, or artistic drive?) In New developments in bioscience: their implications for laboratory animal science. Ed. by A.O. Beynen and H.A. Solleveld. Martinus NijhoffPublishers. pp 305-309.
44. Clarke, A.S., Mason, W.A. and Mendoza, S.P. (1994). Heart rate patterns under stress in three species of macaques. American Journal of Primatology 33:133-148.
45. Boccia, M.L., Laudenslager, M.L. and Reite, M.L. (1995). Individual differences in macaques' responses to stressors based on social and physiological factors: implications for primate welfare and research outcomes. Laboratory Animals 29:250-257.
46. Sackett, G.P., Holm, R.A. and Ruppenthal, G.C. (1976). Social isolation rearing: species differences in behaviour of macaque monkeys. Developmental Psychology 12:283-288.
47. Worlein, J.M. and Sackett, G.P. (1997). Social development in nursery-reared pig-ail macaques (Macaca nemistrina). American Journal of Primatology 41:23- 35.
48. Mason, W.A. and Berkson, G. (1975). Effects of maternal mobility on the development of rocking and other behaviors in rhesus monkeys: a study with artificial mothers. Developmental Psychobiology 8: 197-211. 49. Erwin, J., Mitchell, G. and Maple, T. (1973). Abnormal behavior in non- isolate-reared rhesus monkeys. Psychological Reports 33:515-523.
50. Suomi, S.J., Kraemer, G.W ., Baysinger, C.M. and DeLizio, R.D. (1981). Inherited and experimental factors associated with individual differences in anxious behavior displayed by rhesus monkeys. In Anxiety: new research and changing concepts. Ed. by D.F. Klein and J. Rabkin. Raven Press. New York. pp 179-199.
Reproduced with permission of the Institute of Animal Technology.